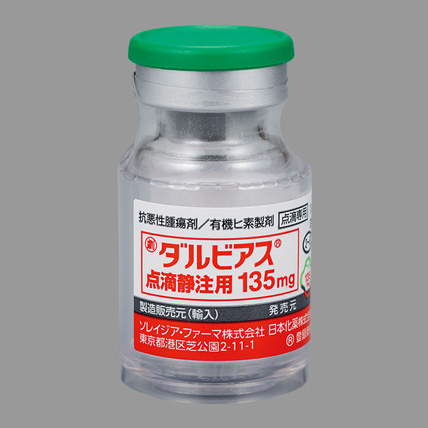
DARVIAS® Injection 135 mg

【Information】
| Trade name | DARVIAS® Injection 135mg |
|---|---|
| Drug Classification | Antineoplastic agent / Organic arsenic product |
| Generic name | Darinaparsin |
| Indications | Relapsed or refractory peripheral T-cell lymphoma (PTCL) |
| Regulatory Category | Powerful drug・Prescription-only drug (Caution - Use it under a prescription of a physician, etc.) |
| Marketing Authorization Holder | Solasia Pharma K.K. (Japan) |
| Commercialization and promotion partner | Nippon Kayaku |
The distributor in Japan is Nippon Kayaku Co. Ltd.
For more information, please link to Nippon Kayaku website below; (only available in Japanese)
https://mink.nipponkayaku.co.jp/product/darvias/
Information for use of our products
The site you are about to access is intended to provide information for medical professionals in Japan. It is designed to allow users to better understand Solasia's products.
I have read and understood above